CSIRO’s biologics facilities play a vital role in the biomedical ecosystem. Our expert scientists and world-class facilities provide companies and other research organisations the opportunity to produce biologics at the scale and quality required for pre-clinical and clinical applications. We make antibodies, antibody conjugates (e.g. theranostics) and vaccines (protein subunit vaccines, viral vector vaccines, virus-like particles) and other biologics and have the necessary accreditation for our partners to evaluate their research in clinical trials.
Working with us – Vaccines and therapeutics
Download the factsheet
- Working with us – vaccines and therapeutics
- Working with us – vaccines and therapeutics (accessible text only)
CSIRO's national biologics laboratories
National Vaccine and Therapeutics Lab (cGMP)
Launching in August 2022, the National Vaccine and Therapeutics Laboratory is a multipurpose facility to produce a variety of biologics, including recombinant proteins and peptides and viral products for therapeutic applications in mammalian and insect cells. The facility operates under cGMP regulations enabling production of biologics for clinical trials (Phase I and II).
It enables process development and production of clinical-grade biologics (protein drugs and vaccines) to address an unmet demand in the Australian Biotech landscape.
This new facility is designed and operated to be compliant with Australia’s Therapeutic Goods Administration (TGA), United States Federal Drug Administration (US FDA) and European Medicines Agency (EMA) regulations. This means that the Australian Biotech industry will have access to global clinical trial opportunities and a broader, faster path to market.
NCRIS Mammalian Biologics Facility
Our Biologics Production Facility is a catalyst for biotechnology research and innovation in Australia and supports Australian biosecurity and health.
Our facility is part of the National Collaborative Research Infrastructure Strategy program and enables us to:
- produce biologics in mammalian and insect cell cultures as well as purify them in a clean room – run under a Quality Management System and is ISO9001 and Veterinary cGMP certified
- support therapeutic discovery with optimisation, scale‑up, production and purification of recombinant protein drugs and vaccines in large quantities – from hundreds of milligrams to tens of grams
- manufacture proteins for conjugation for imaging and tissue targeting drugs.
NCRIS Microbial Research and Development Facility
CSIRO’s Microbial Research and Development Facility is also part of the NCRIS program and offers process development and pilot-scale demonstration of processes used to manufacture microbial products with therapeutic applications.
The facility provides key infrastructure and skills enabling therapeutic products to transition toward commercial realisation. To support this the facility has:
- Multiple parallel bioreactors for rapid process development
- A high degree of flexibility to cater for many different microbial products and process configurations
- Pilot-scale process demonstration up to 400 litres
- Downstream processing suitable for different microbial products at both small- and pilot-scale.
QC Laboratories
With construction underway shortly, our purpose‑built Quality Control (QC) Laboratory will provide process testing services to support the manufacturing and release of biologics produced in the cGMP Biologics Facility and the NCRIS Mammalian Biologics Facility.
It will also provide local, accessible, and bespoke solutions for Australian companies and researchers engaging in early‑stage development of protein drugs and vaccines. Services include testing for: material quality, in process quality, end‑product quality release, stability testing, potency testing and development of bespoke potency assays and drug product stability testing.
CSIRO’s biologics facilities play a vital role in the biomedical ecosystem. Our expert scientists and world-class facilities provide companies and other research organisations the opportunity to produce biologics at the scale and quality required for pre-clinical and clinical applications. We make antibodies, antibody conjugates (e.g. theranostics) and vaccines (protein subunit vaccines, viral vector vaccines, virus-like particles) and other biologics and have the necessary accreditation for our partners to evaluate their research in clinical trials.
Working with us – Vaccines and therapeutics
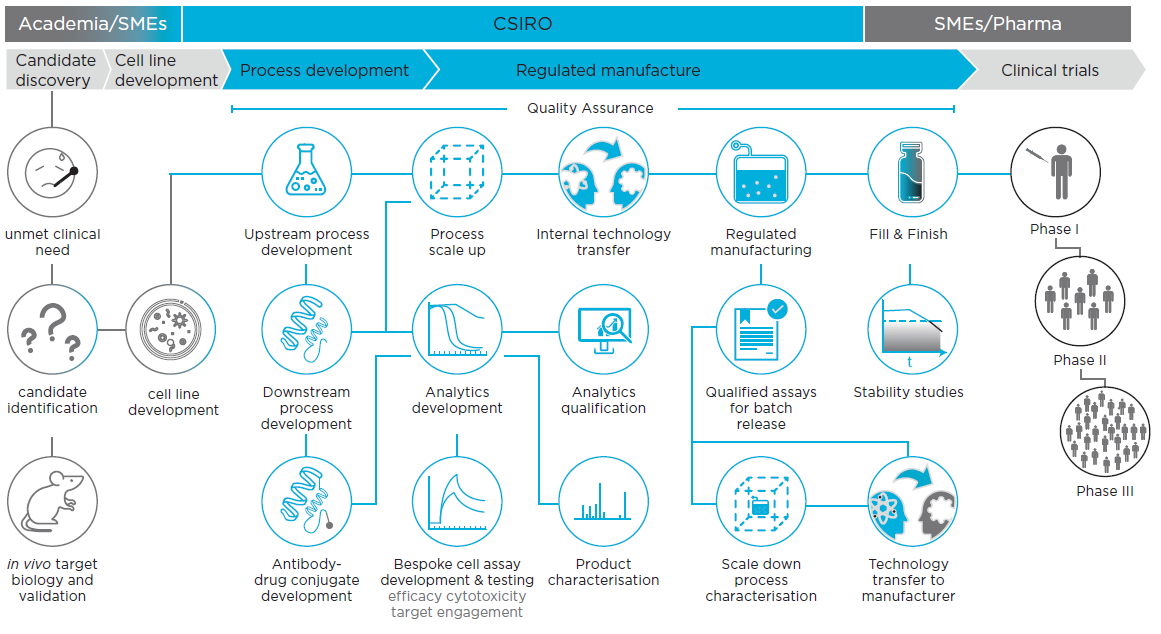
unmet clinical
need
in vivo target
biology and
validation
Process development
cell line
development
Upstream process
development
Downstream
process
development
Analytics
development
Analytics
qualification
Qualified assays
for batch
release
Bespoke cell assay
development & testing
efficacy cytotoxicity
target engagement
Product
characterisation
Scale down
process
characterisation
Technology
transfer to
manufacturer
Stability studies
Antibodydrug conjugate
development
Process
scale up
Internal technology
transfer
Regulated
manufacturing
Fill & Finish
Cell line
development
Candidate
discovery
Phase I
Phase II
Phase IIIWorking with us –
Vaccines and therapeutics
Download the factsheet
- Working with us – vaccines and therapeutics PDF (312 KB)
- Working with us – vaccines and therapeutics (accessible text only) TXT (3 KB)
CSIRO's national biologics laboratories
National Vaccine and Therapeutics Lab (cGMP)
Launching in August 2022, the National Vaccine and Therapeutics Laboratory is a multipurpose facility to produce a variety of biologics, including recombinant proteins and peptides and viral products for therapeutic applications in mammalian and insect cells. The facility operates under cGMP regulations enabling production of biologics for clinical trials (Phase I and II).
It enables process development and production of clinical-grade biologics (protein drugs and vaccines) to address an unmet demand in the Australian Biotech landscape.
This new facility is designed and operated to be compliant with Australia’s Therapeutic Goods Administration (TGA), United States Federal Drug Administration (US FDA) and European Medicines Agency (EMA) regulations. This means that the Australian Biotech industry will have access to global clinical trial opportunities and a broader, faster path to market.
NCRIS Mammalian Biologics Facility
Our Biologics Production Facility is a catalyst for biotechnology research and innovation in Australia and supports Australian biosecurity and health.
Our facility is part of the National Collaborative Research Infrastructure Strategy program and enables us to:
- produce biologics in mammalian and insect cell cultures as well as purify them in a clean room – run under a Quality Management System and is ISO9001 and Veterinary cGMP certified
- support therapeutic discovery with optimisation, scale‑up, production and purification of recombinant protein drugs and vaccines in large quantities – from hundreds of milligrams to tens of grams
- manufacture proteins for conjugation for imaging and tissue targeting drugs.
NCRIS Microbial Research and Development Facility
CSIRO’s Microbial Research and Development Facility is also part of the NCRIS program and offers process development and pilot-scale demonstration of processes used to manufacture microbial products with therapeutic applications.
The facility provides key infrastructure and skills enabling therapeutic products to transition toward commercial realisation. To support this the facility has:
- Multiple parallel bioreactors for rapid process development
- A high degree of flexibility to cater for many different microbial products and process configurations
- Pilot-scale process demonstration up to 400 litres
- Downstream processing suitable for different microbial products at both small- and pilot-scale.
QC Laboratories
With construction underway shortly, our purpose‑built Quality Control (QC) Laboratory will provide process testing services to support the manufacturing and release of biologics produced in the cGMP Biologics Facility and the NCRIS Mammalian Biologics Facility.
It will also provide local, accessible, and bespoke solutions for Australian companies and researchers engaging in early‑stage development of protein drugs and vaccines. Services include testing for: material quality, in process quality, end‑product quality release, stability testing, potency testing and development of bespoke potency assays and drug product stability testing.
